

Calculate your potential savings with our ROI Calculator
ROI CalculatorKeep your core quality processes including CAPA, nonconformance, deviations, audits, supplier quality, document control, incidents, and training connected in one closed-loop system.
Trusted by global leaders across regulated industries like Life Sciences, Medical Devices, Pharmaceuticals, Manufacturing, Automotive, Aerospace, and Healthcare.


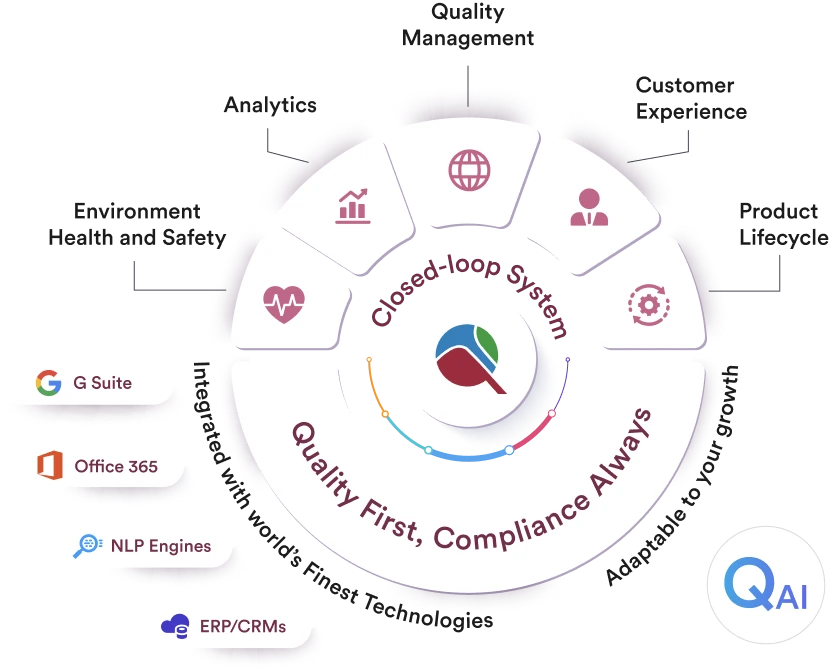
Qualityze unify your data, workflows, and teams on a centralized platform, giving you full visibility and traceability across the lifecycle of every quality event.
Simple. Scalable. Designed for modern quality teams.
Qualityze next-generation EQMS and EHS solutions help regulated teams build repeatable standard best-practices workflows around approvals, reviews, training, documents and change control to keep quality and safety in control even when no one is watching.
Secure. 100% Verified. Ready for auditor’s review always
Life Sciences, Medical Devices, Pharmaceuticals, Manufacturing, Automotive, Aerospace, Healthcare
See how Qualityze intelligent quality solutions help teams to speed up investigations, closures, and data-driven decisions with a dedicated agent for every core process including NC, CAPA, Training, Incident, Complaints, and more.
Our specialized QAI agents help your teams to:
Qualityze Intelligence brings clarity, speed, and innovation it takes for regulated environments to continuously improve Delivered Quality .
Because we bring best-in-class quality, safety, and compliance-oriented solutions for Life Sciences, Medical Devices, Manufacturing, Automotive, and other compliance-controlled industries to simplify everyday quality processes.
Having delivered more than 20 solutions spanning QMS and EHS categories with specialized QAI agents, Qualityze remains committed to excellence when it comes to delivering value to people who prioritize quality.
Improved Compliance
Faster audits and inspections
Reduced operational costs
Enhanced product quality and safety
When we say Qualityze Difference, it generally is about the decade of experience and expertise woven into an entire range of smarter quality solutions. Each solution has intuitive dashboard, guided navigation into workflows from initiation through closure, and contextual help to make quality management simple for every user.
Industry leading enterprises choose Qualityze for:
Qualityze is built to support this vision. Our intelligent quality solutions helps teams migrate from legacy tools to one connected suite that helps them to;
Focus on Metric That Matters for Everyone: Delivered Quality






Ready for a real talk moment? We often see compliance as a necessary evil that comes with...
Quality is how well something meets its intended purpose and satisfies customer expectatio...
Maintaining the highest standards of quality in an ever-evolving landscape of industries l...
ISO and QMS: An Enterprises Perspective Businesses commonly standardize their processes. T...