

Calculate your potential savings with our ROI Calculator
ROI CalculatorReduce nonconformance management cycle times while increasing control and visibility over every quality issue reported. Our AI powered nonconformance software helps eliminate process inefficiencies by automating workflows, ensuring regulatory compliance, and providing real-time insights that empower your team to investigate and resolve issues faster.
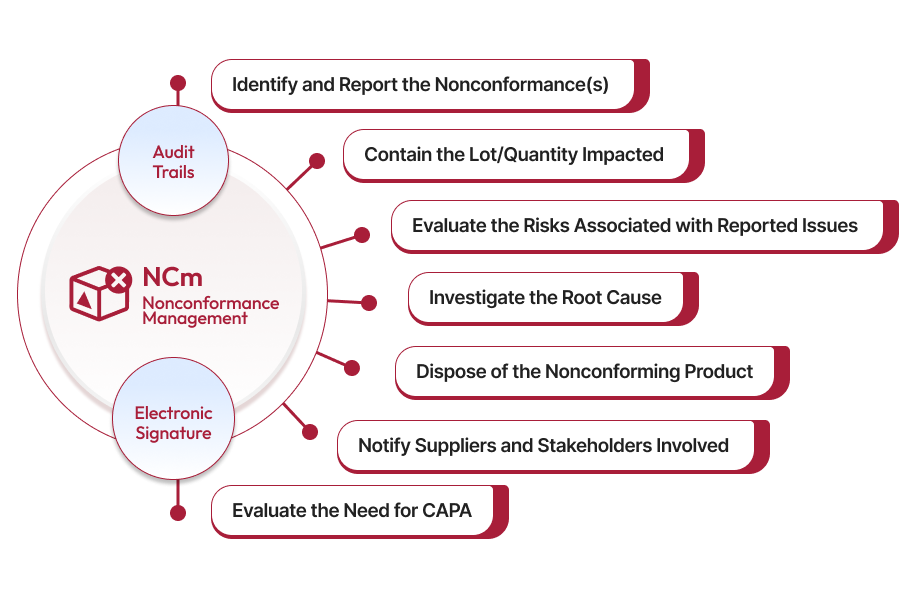
Detect patterns and predict issues before they escalate with our QAI assistant that provides real-time insights and automates routine tasks for faster resolutions like root cause analysis, suggests corrective actions, and recommends next-steps to accelerate your nonconformance workflows, ensuring timely resolutions.
If you have more questions feel free to reachout to us.
Contact UsNonconformance Management Software is a system that helps businesses detect, document, and resolve nonconformance events that deviate from quality standards. It automates workflows, tracks issues, and ensures compliance with regulatory standards, helping organizations resolve quality problems efficiently and prevent them from recurring.
Qualityze offers a comprehensive nonconformance management system that automates the entire process, from detection and categorization to root cause analysis and corrective actions. It provides real-time tracking, automated workflows, and AI-driven insights to streamline nonconformance management and ensure regulatory compliance.
While nonconformance management focuses on identifying and resolving quality issues that have already occurred, CAPA involves implementing corrective actions to fix the root cause of the issue and preventive actions to stop it from happening again in the future. Nonconformance management is often the starting point for initiating CAPA.
Defect code management is critical in a non conformance tracking system because it standardizes how issues are categorized, making it easier to track recurring problems, analyze trends, and implement corrective actions. By using predefined or customizable defect codes, businesses can ensure consistent categorization and faster resolution of nonconformance events.
Nonconformance management software is widely used in industries with stringent quality and regulatory requirements, such as manufacturing, healthcare, pharmaceuticals, medical devices, automotive, and aerospace. These industries rely on nonconformance management to ensure product quality, safety, and compliance with standards like ISO and FDA regulations.
AI enhances nonconformance management by automating manual tasks, detecting patterns in nonconformance events, and predicting future quality risks. AI-powered insights help organizations resolve issues faster and prevent similar nonconformances from recurring. Qualityze AI-powered recommendations make it easier to identify root causes, implement corrective actions, and continuously improve quality management processes.
A compliant nonconformance tracking system must align with industry regulations such as FDA, ISO, and GxP. It should provide features like audit-ready documentation, traceability, automated workflows, and reporting capabilities. Qualityze ensures compliance with built-in regulatory checks, documentation features, and audit trails.
Root Cause Analysis is a crucial part of nonconformance management, helping to identify the underlying reasons for a nonconformance event. Tools like 5 Whys or Fishbone Diagrams are commonly used to determine why the issue occurred, ensuring that corrective actions target the real problem and preventing its recurrence.
Qualityze provides a robust, cloud-based nonconformance tracking system that offers real-time visibility into nonconformance events. With customizable dashboards, automated notifications, and integrated reporting tools, Qualityze ensures that teams can track, manage, and resolve nonconformances in real-time, minimizing downtime and enhancing overall quality management.
Qualityze EQMS ensures compliance by providing features that comply with industry-specific standards, such as ISO 9001, ISO 13485, 21 CFR Part 11, 21 CFR Part 820, AS9100, and IATF 16949. The solution is built on the Salesforce platform, which provides a secure and compliant cloud infrastructure. Qualityze EQMS allows businesses to maintain a complete audit trail of all quality data, ensuring regulatory compliance and reducing the risk of non-compliance.The software also provides configurable workflows that allow businesses to enforce compliance with their own internal policies and procedures. The workflows can be customized to fit specific requirements and ensure that all processes are standardized and consistent across the organization. The solution provides real-time visibility and reporting, allowing businesses to quickly identify areas of non-compliance and take corrective action. Qualityze EQMS also provides automatic version control and document management features, ensuring that all documents are up-to-date and comply with relevant regulations.
As a cloud-based solution, Qualityze EQMS is continually updated with the latest features and functionalities to ensure compliance with changing regulations and industry standards. Qualityze provides free lifetime updates to its customers to keep them up-to-date with the latest technology advancements and features. These updates are automatically applied to the system without any additional cost or disruption to the user. Qualityze’s software as a service (SaaS) model allows for seamless integration of new features and bug fixes, which means that customers always have access to the latest version of the software. Qualityze’s dedicated team of developers and quality experts continuously work on enhancing the product to ensure that it meets the evolving needs of the customers and helps them stay ahead of the competition.
Qualityze's free lifetime updates include bug fixes, security updates, feature enhancements, and new functionalities. These updates ensure that the Qualityze EQMS solution stays up-to-date and continues to meet the evolving needs of the industry and regulatory standards. The updates are provided without any additional cost to the customers and are automatically deployed to their systems. The Qualityze team ensures that the updates are thoroughly tested before being released to the customers to ensure their smooth integration and minimal disruption to their business operations. The free lifetime updates help customers to leverage the latest technological advancements and ensure that their quality management processes remain compliant and effective.
Qualityze QMS provides a highly secure environment for managing quality data. It is built on the world’s leading cloud-based platform, Salesforce.com, which has a robust security framework with multiple layers of security features, including:
Overall, Qualityze QMS provides a secure environment for managing quality data, giving organizations peace of mind that their sensitive data is protected.